Plastics and microplastics
My research with the ADRIFT Lab focuses on understanding how plastic consumption harms wildlife, from visible tissue damage through to the molecular signals that predict disease. Working across pathology, histology, and biomarker discovery, we examine how ingested plastics interact with the gut and internal organs to trigger inflammation, scarring, and disrupted tissue function. We pair classical pathology (gross examinations, histopathology, lesion scoring, and organ-level assessment) with molecular approaches such as proteomics and targeted marker panels to identify the biological pathways most affected by plastic exposure.
Publications
Charlton-Howard, H. S., Bond, A. L., Rivers-Auty, J., & Lavers, J. L. (2023). ‘Plasticosis’: Characterising macro- and microplastic-associated fibrosis in seabird tissues. Journal of hazardous materials, 450, 131090. https://doi.org/10.1016/j.jhazmat.2023.131090
Keys, B. C., Grant, M. L., Rodemann, T., Mylius, K. A., Pinfold, T. L., Rivers-Auty, J., & Lavers, J. L. (2023). New Methods for the Quantification of Ingested Nano- and Ultrafine Plastics in Seabirds. Environmental science & technology, 57(1), 310–320. https://doi.org/10.1021/acs.est.2c06973
Rivers-Auty, J., Bond, A. L., Grant, M. L., & Lavers, J. L. (2023). The one-two punch of plastic exposure: Macro- and micro-plastics induce multi-organ damage in seabirds. Journal of hazardous materials, 442, 130117. https://doi.org/10.1016/j.jhazmat.2022.130117
de Jersey, A. M., Lavers, J. L., Zosky, G. R., & Rivers-Auty, J. (2023). The understudied global experiment of pollution’s impacts on wildlife and human health: The ethical imperative for interdisciplinary research. Environmental pollution (Barking, Essex : 1987), 336, 122459. https://doi.org/10.1016/j.envpol.2023.122459
Mylius, K. A., Lavers, J. L., Woehler, E. J., Rodemann, T., Keys, B. C., & Rivers-Auty, J. (2023). Foraging strategy influences the quantity of ingested micro- and nanoplastics in shorebirds. Environmental pollution (Barking, Essex : 1987), 319, 120844. https://doi.org/10.1016/j.envpol.2022.120844
Jones, N. R., de Jersey, A. M., Lavers, J. L., Rodemann, T., & Rivers-Auty, J. (2024). Identifying laboratory sources of microplastic and nanoplastic contamination from the air, water, and consumables. Journal of hazardous materials, 465, 133276. https://doi.org/10.1016/j.jhazmat.2023.133276
Dementia and Ageing Research
My dementia research spans three complementary scales: whole organisms, cell biology, and population-level epidemiology. Using animal models, I investigate how immune dysregulation and chronic inflammation shape cognitive decline and neurodegenerative pathology, with a focus on the pathways that convert an initially protective inflammatory response into one that becomes maladaptive and tissue-damaging over time. These models allow us to test causality and to explore how environmental and metabolic stressors can accelerate disease-relevant phenotypes.
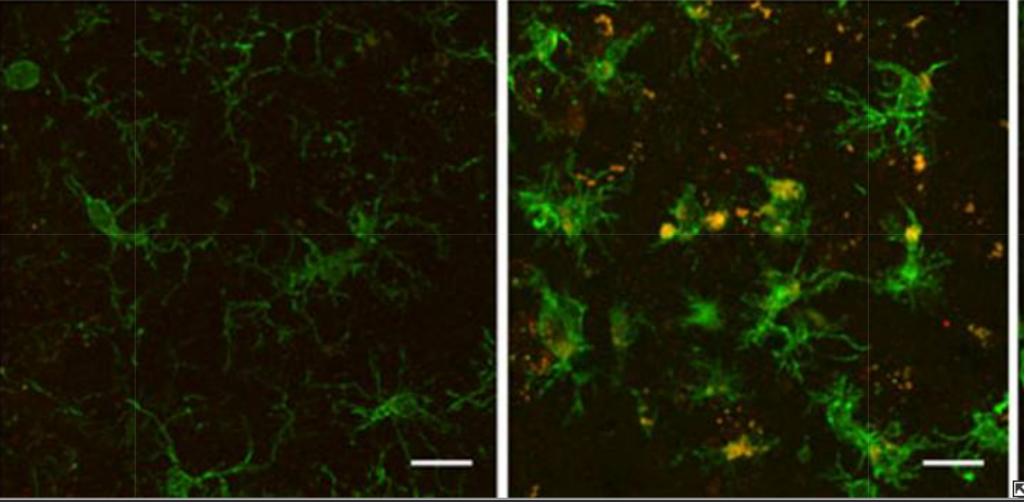
At the cellular level, my work examines how dementia-related protein aggregates interact with neurons and glial cells, particularly astrocytes and microglia. I am interested in how these aggregates are handled, how they propagate stress signals, and how innate immune signalling can amplify neuronal dysfunction. This includes probing inflammatory mediators and upstream triggers that prime or activate microglia, and identifying molecular signatures that distinguish adaptive clearance and repair from harmful, self-sustaining neuroinflammation.
In parallel, I apply epidemiological methods to human health data to understand how these mechanisms translate to real-world risk and trajectories. Using longitudinal and time-series approaches, I study patterns of cognitive decline and the factors that modify them, including inflammation-related exposures and medication use. By integrating mechanistic insights from animal and cellular work with epidemiological evidence, my research aims to produce a coherent, translational picture of dementia risk and progression, and to identify practical intervention points that can be tested experimentally and evaluated at scale.
Publications
White, C. S., Lawrence, C. B., Brough, D., & Rivers-Auty, J. (2017). Inflammasomes as therapeutic targets for Alzheimer’s disease. Brain pathology (Zurich, Switzerland), 27(2), 223–234. https://doi.org/10.1111/bpa.12478
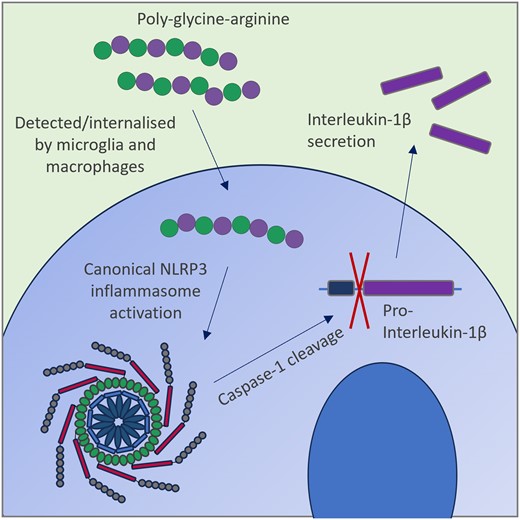
Rivers-Auty, J., Hoyle, C., Pointer, A., Lawrence, C., Pickering-Brown, S., Brough, D., & Ryan, S. (2024). C9orf72 dipeptides activate the NLRP3 inflammasome. Brain communications, 6(5), fcae282. https://doi.org/10.1093/braincomms/fcae282
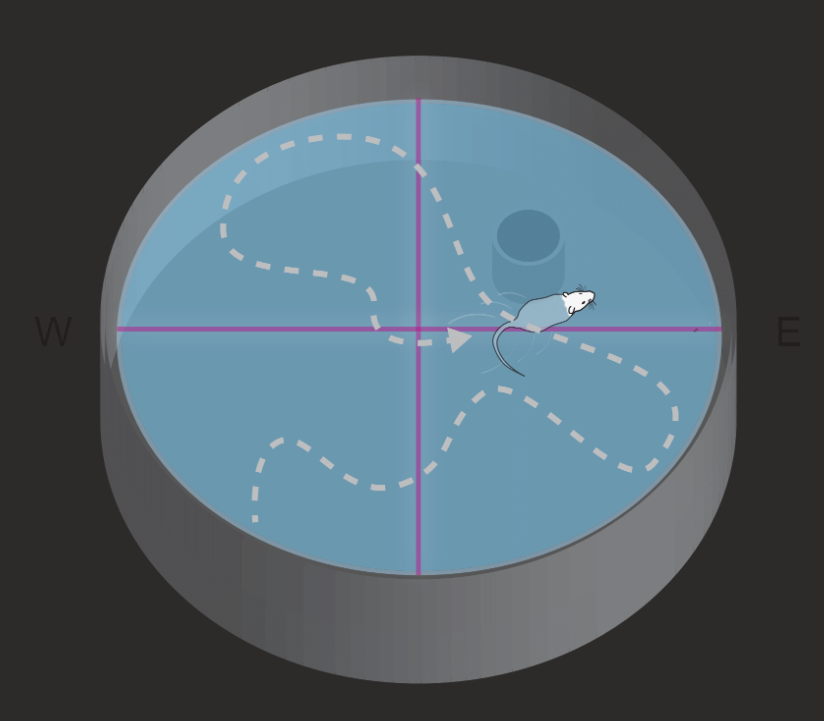
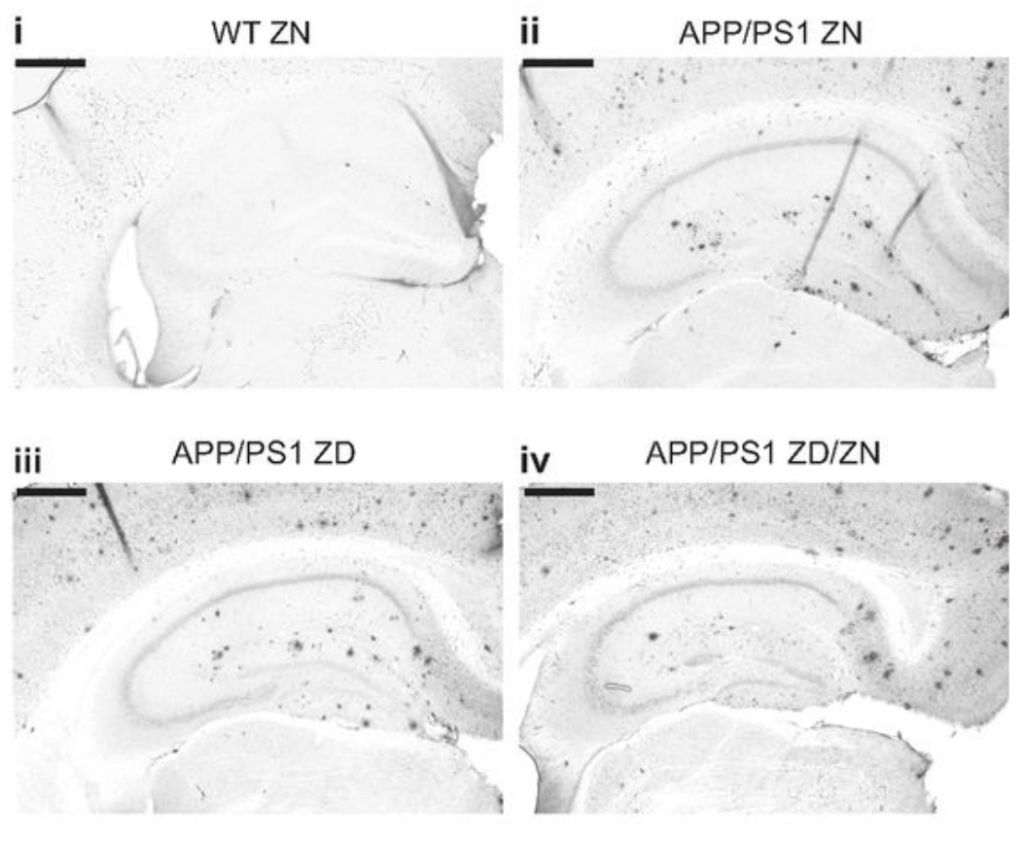
Rivers-Auty, J., Tapia, V. S., White, C. S., Daniels, M. J. D., Drinkall, S., Kennedy, P. T., Spence, H. G., Yu, S., Green, J. P., Hoyle, C., Cook, J., Bradley, A., Mather, A. E., Peters, R., Tzeng, T. C., Gordon, M. J., Beattie, J. H., Brough, D., & Lawrence, C. B. (2021). Zinc Status Alters Alzheimer’s Disease Progression through NLRP3-Dependent Inflammation. The Journal of neuroscience : the official journal of the Society for Neuroscience, 41(13), 3025–3038. https://doi.org/10.1523/JNEUROSCI.1980-20.2020
Hadjidemetriou, M., Rivers-Auty, J., Papafilippou, L., Eales, J., Kellett, K. A. B., Hooper, N. M., Lawrence, C. B., & Kostarelos, K. (2021). Nanoparticle-Enabled Enrichment of Longitudinal Blood Proteomic Fingerprints in Alzheimer’s Disease. ACS nano, 15(4), 7357–7369. https://doi.org/10.1021/acsnano.1c00658
Daniels, M. J., Rivers-Auty, J., Schilling, T., Spencer, N. G., Watremez, W., Fasolino, V., Booth, S. J., White, C. S., Baldwin, A. G., Freeman, S., Wong, R., Latta, C., Yu, S., Jackson, J., Fischer, N., Koziel, V., Pillot, T., Bagnall, J., Allan, S. M., Paszek, P., … Brough, D. (2016). Fenamate NSAIDs inhibit the NLRP3 inflammasome and protect against Alzheimer’s disease in rodent models. Nature communications, 7, 12504. https://doi.org/10.1038/ncomms12504